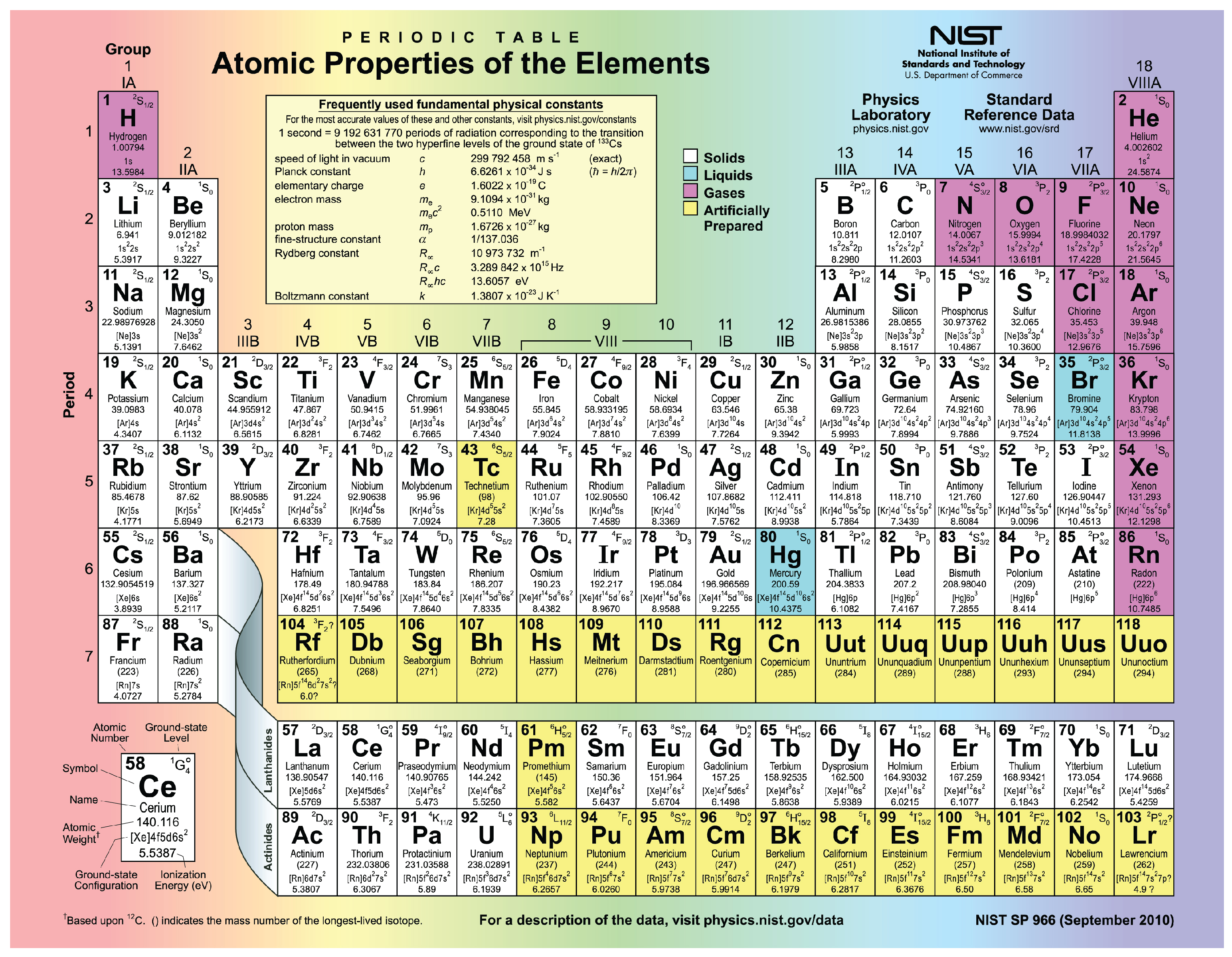
For example, a quick glance at Figure AT5.2. That way, we can get a better look at the relationship. Electronegativity decreases down a column of the. Often it is useful to plot data on a graph. Determine the number of valence electrons in neutral atoms of the following elements. As a result, fluorine is usually thought of as the most electronegative element. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number i.e., the total number of protons in the atomic nucleus. However, on many scales, fluorine would be the most electronegative atom here. As a result, noble gases are also given electronegativity values on this scale. The Allen scale just depends on the ability of an atom to interact with light, which is something even noble gases can do. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. This table shows the pattern in the periodic table that Mendeleev developed and how the missing elements at. Some electronegativity scales do not have values for the noble gases, because they are based on experimental measurements of compounds, and noble gases do not commonly form compounds with other elements. Electrons orbit the atoms nucleus in energy levels. Element C is located in Period 5, the 1st position in 5s. Not every atom has the same number of valence electrons, it depends on the location of the element on the periodic table. group I group VI group III, Using the periodic table. What is its ionic charge, Using the periodic table, determine the ion charges of the following families of elements if valence electrons were removed or added to reach the octet. This means that B has two valence electrons in 3s (3s 2). Periodic table with valence electrons is shown in the above image. Study with Quizlet and memorize flashcards containing terms like An element of group II loses two electrons in the process of a chemical combination. Element B is located in Period 3, the 2nd position in 3s-block. \): The Allen electronegativity values of the second-row elements. This means that A has two valence electrons in 2s (2s 2) and five valence electrons in 2p (2p 5).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
